|
12/12/2023 0 Comments Ag charge ion 
Putting the above in an acid will add more H +, and thus, drive more B to dissolve according to Le Chatelier's principle.Putting the above in a base will take out the H +, thus, more HA will dissolve according to Le Chatelier's principle.Here, the complex ion formation takes out Ag +, again causing more AgCl to dissolve.Silver most commonly occurs in two oxidation states (Ag0, Ag+) and it. Alternatively: AgCl (s) ↔ Ag + (aq) + Cl - (aq) NH 3 + Ag + ↔ Ag-(NH 3) n complex ion. The toxicity of the silver ion is well studied (Ratte, 1999 Hogstrand and Wood, 1998).When complex ion forms, the Cl - ion is taken out, so more of the AgCl will dissolve.AgCl (s) ↔ Ag + (aq) + Cl - (aq) M + + Cl - ↔ M-Cl n complex ion.The "complex ion effect" is the opposite of the common ion effect.The K eq for this reaction is called K f, or the formation constant.The Lewis base can be charged or uncharged.This will selectively crash out AgCl by the common ion effect (Cl - being the common ion). For example, if you want to separate AgCl from a mixture of AgCl and Ag 2SO 4, then you can do so by adding NaCl.In laboratory separations, you can use the common ion effect to selectively crashing out one component in a mixture.Another example: more AgCl can dissolve in pure water than in water containing Cl - ions.For example, if you add NaCl to a saturated solution of AgCl, then some AgCl will crash out of solution.The common-ion effect says that if you add Cl - to the solution above, then less AgCl would dissolve.But you need to know the specific ionic charge elments. Non-metals (found on the right) will be negative. On the Periodic Table metals (found on the left of the table) will be positive. To find the ionic charge of an element youll need to consult your Periodic Table. The common-ion effect is simply Le Chatelier's principle applied to K sp reactions. Finding Charges of Ions on Periodic Table. 
Common-ion effect, its use in laboratory separations.If you were given solubility and asked to solve K sp, then know that solubility = /2.If you solved for instead, divide your results by 2.Solubility is the same thing as because you used Q = K sp for a saturated solution. K sp = 2 (because for every M 2+, there's two times as much X -).What is the solubility of MX 2 if given K sp?.The higher the K sp, the more the reaction products dominate in a saturated solution (at equilibrium).Solubility product constant, the equilibrium expression.x ppm = x parts per million = x mg / kg = x mg / L.Give the names of the anion in each of the following compounds: CuS, Ag2SO4, Al (ClO3)3, Co (OH)2, PbCO3. Give the charges of the cation in each of the following compounds: CuS, Ag2SO4, Al (ClO3)3, Co (OH)2, PbCO3. Normality = N = Molarity of the species that matter. Give the names of the cation in each of the following compounds: CuS, Ag2SO4, Al (ClO3)3, Co (OH)2, PbCO3. 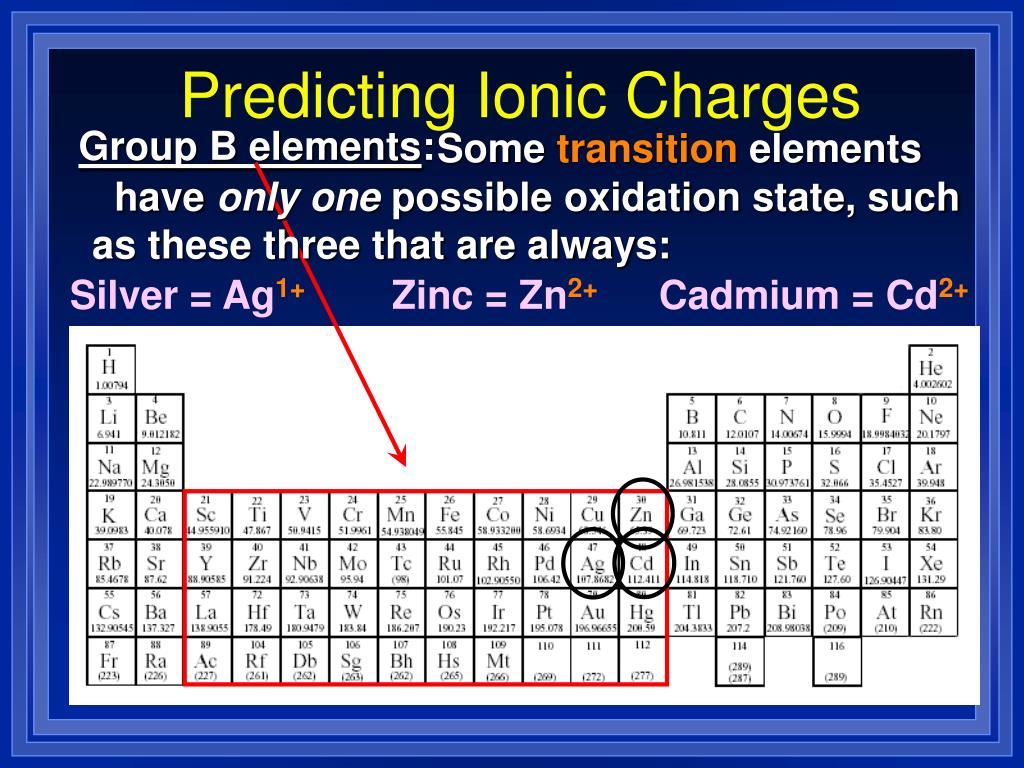
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
